
Today is the day. And I'm pretty excited about it!
I intended to get out of bed around 7. I dismissed my 3 or 4 alarms and rolled over to see not even the faintest beams of light through my shades. I allowed myself 20 more minutes. “Today is the day,” I thought. Today is the day I nail it. My very last go-round in the endocrine lab. I don’t think I’ve ever ran an assay and had every sample come out successfully. But this is not my first or second time with these particular samples. No. This is the third time for most of them. I slipped into my slippers, greeted my dog on the way to the kitchen, and poured myself a glass of water to wash down the ball of sunshine my body is deficient of for about 5-6 months out of the year. I put together a cold sandwich. The Dean is visiting our campus today and a student lunch is provided but the chances of me pipetting over the lunch hour are too high to risk going without. I reach for the coffee pot, second in line of the joys of dark mornings (first being the thick ruff of my Hedlund husky that I scratch and bury my face into each morning). Here I come to a cross-roads. In the summer I could go on my merry way without the dark liquid. But on a frosty December morning such as this, my day’s happiness and productiveness depend on it. But I must be cautious. No one wants the shakes when they are transferring microliters of precious samples that have traveled hundreds of miles and gone through a dozen steps to be dropped anywhere outside the pencil eraser-sized cup (a “well”) in the assay tray. I settle on a half cup.
A row of "wells."
I run out of the house to start my car. Two days ago I opened my back door and it resisted, so I cranked a little harder. The entire panel of my passenger side back door popped off. I threw it in the back. Today, my driver’s side door is frozen shut. I have to crawl through the back. I can’t be bothered to take this thing in as long as it’s getting me from point a to point b.
Half an hour later I’m in the lab rooting around in the fridge. It’s jam packed with blue and yellow boxes labeled, “testosterone,” “cortisol,” and “corticosterone.” I remove a few of them to get to the one labeled “progesterone.” A beaker of clear liquid labeled “fecal extraction” wobbled precariously. I hadn’t noticed it there. I would like to say my lab mates should know better than to leave something like that balancing in the fridge, but the truth is, I should know them better. I riffle through the progesterone kit and begin my hodge-podge assembled of my assay, pulling out buffer from the side door in the main fridge, grabbing the control solution from the mini fridge, and piecemealing together unused well columns from a recycled tray. Often we don’t have enough samples to use up an entire assay plate, so we save and label the leftovers and use them up later. Kits are costly, about $250 for each individual progesterone kit. Conserving materials is wise and limiting mistakes is necessary!
An assay tray.
Sample prep. Thawing out my dilutions and allowing the assay buffer to thaw to room temperature.
A couple hours later and I have my fingers crossed that this assay will be a success. I have been running these damn things on and off for more than a year. Some, because we obtained new samples (wahoo!), but mainly they are redos that I must recalculate over and over again. You see the trouble is no one has done this before, ran progesterone on walrus tissues. And these aren’t just any old piece of tissue (the skin, blubber etc.); the part of the ovary I am observing (the Corpora Lutea or CL) is the source of progesterone production itself. I have yet to find literature looking at progesterone concentration in the CL of any mammal. This makes it tricky to blindly guess. Luckily for me, our lab has an excellent technician who’s been invaluable at assisting students in endocrinology studies. But today I am on my own.
Plate is ready to shake!
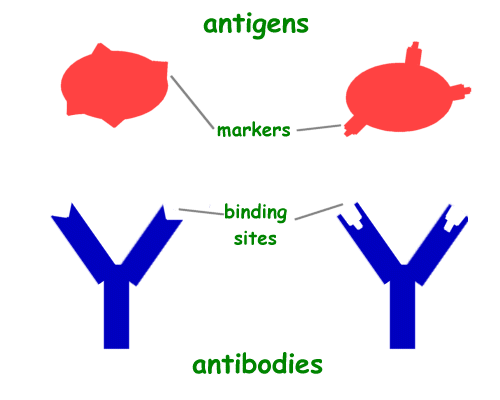
The plate shakes and by some miracle the 2-hour time window of shaking falls during the student lunch! Yay for Chinese food! Of course, the food arrives late and the Dean is behind schedule so I only have enough time to dine and ditch before time is up and I must complete the next step. In the next step I dump all of the liquid out of the wells and wash them out with something similar to a detergent. This step gets rid of all the “junk” floating around that isn’t progesterone. The progesterone molecules (antigens really) have spent the past 2 hours binding to their respective antibodies. The antibodies come with the kit. They are stuck to the bottom of those tiny cups we call wells and sit upright, arms wide open, waiting for the progesterone antigens to bind to them.
The blue antibodies are stuck to the bottom of the wells, the red antigens are the progesterone molecules from my walrus tissues floating around. They will bind to the antibodies. Source: http://www.ducksters.com/science/biology/antibodies_and_antigens.gif
The plate is washed, I follow the chemical recipe and now the plate is sitting. No shaking for this last part. I am anxiously waiting. I don’t allow any negative thoughts or potential mistakes to enter my mind. I look down at the fortune cookie I saved from lunch. Nope. I’m not even going to open that, could be some bad mojo in there. I’m acting more like some superstitious baseball pitcher before playoffs than a scientist. Instead of letting my mind wander in all the wrong directions, I let my dog out and catch some air. I’ll head up early to prepare for the final steps.
Check out this video on what it looks like to "wash the plate."
0 Comments
Leave a Reply. |
AuthorHi, I'm Jenell. Biologist, student, outdoors woman and author of Mother Walrus. Archives
March 2019
Categories |




 RSS Feed
RSS Feed
